Butyrate / butyric acid and cancer
Summary about butyrate / butyric acid
Effect:
- Butyrate is a short-chain fatty acid produced by our gut bacteria when we consume dietary fiber. It functions as fuel for intestinal cells, has a strong anti-inflammatory effect, and can ‘turn on’ genes in cancer cells that slow their growth.
Potential in cancer:
- Butyrate shows great potential in inhibiting the development of cancer, especially colorectal cancer. It can induce cancer cells to commit suicide (apoptosis), stop their division, and make them more sensitive to chemotherapy and immunotherapy, while protecting healthy intestinal cells.
Main limitation:
- The greatest challenge is that butyrate is rapidly broken down in the body, making it difficult to achieve high concentrations outside the gut through standard supplements. The best source is therefore a fiber-rich diet.
What is butyrate
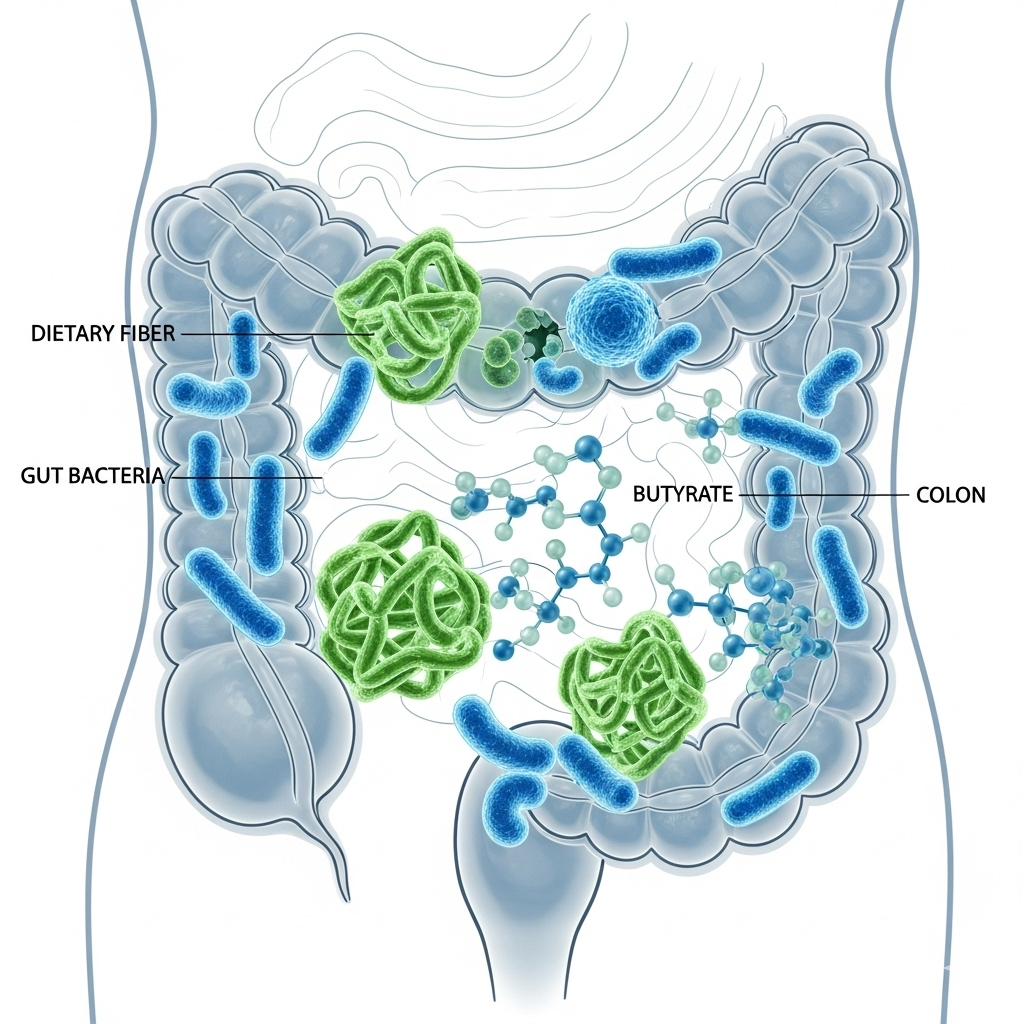
Butyrate, also known as butyric acid, is a short-chain fatty acid (SCFA) that plays a central role in the health of the gut and the entire body. It is not something typically consumed directly in large quantities through the diet, but it is produced naturally in the large intestine.
This production occurs when beneficial gut bacteria ferment (break down) indigestible dietary fibers—particularly resistant starch, found in foods such as unripe bananas, legumes, whole grains, cooled potatoes, and rice.
Butyrate is the primary energy source for the cells in the intestinal wall (colonocytes) and is essential for maintaining a healthy and strong intestinal barrier.
Beyond its local function in the gut, butyrate also has systemic effects, as it is absorbed into the bloodstream and influences processes in the rest of the body, including immune regulation and the cell cycle.
Its unique properties make it an interesting molecule in connection with the prevention and supplementary treatment of cancer, especially colorectal cancer.
History
Butyric acid was discovered in the 1800s in rancid butter and was long regarded as a simple fat. Only in recent decades has research revealed that it is a crucial signaling molecule produced by gut bacteria from dietary fiber.
Modern understanding has shifted the focus from merely seeing butyric acid as energy for intestinal cells to recognizing its profound effects on the body’s health and disease processes.
Mechanisms of action
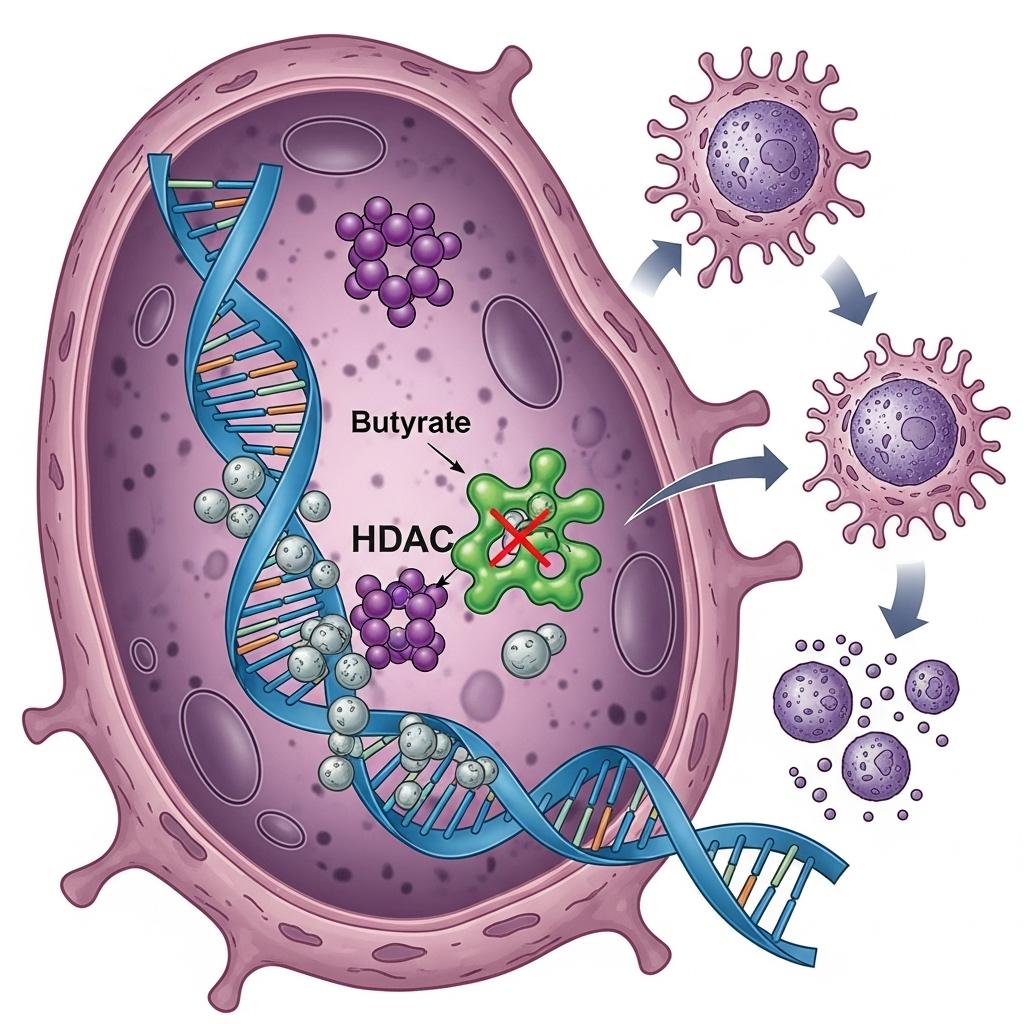
Butyrate’s potential in cancer treatment is due to its ability to influence cancer cells at several fundamental levels. Its mechanisms of action involve both genetic and metabolic processes.
Inhibition of HDAC enzymes
In many cancer cells, important defense genes that control cell growth and cell death (apoptosis) have been ‘turned off’ by overactive HDAC enzymes. Butyrate functions as an HDAC inhibitor: it blocks these enzymes so that protective genes can be ‘turned on’ again. This ability to ‘turn on’ the cell’s silenced defense genes can force the cancer cell to stop its growth or undergo apoptosis [1].
Restoration of cell death (apoptosis)
Butyrate restores the ability of cancer cells to undergo apoptosis. This occurs partly by changing the balance between proteins that promote and inhibit cell death, and partly by making cancer cells more sensitive to the immune system’s killer signals, e.g., via the substance TRAIL (The immune system’s killer signal) [2].
Blocking growth signals and energy supply
Butyrate can slow the growth and spread of cancer cells in two ways. Firstly, it can turn off the internal signals within the cancer cell that function as an accelerator for uncontrolled cell division [3]. Secondly, it attacks the energy supply of cancer cells by limiting their uptake of sugar (glucose), thereby starving them of the fuel they need to grow [3].
Anti-inflammatory effect
Butyrate has strong anti-inflammatory properties, especially in the gut. It works by inhibiting the central inflammatory signaling pathway NF-κB, creating a less favorable environment for the growth and survival of cancer cells [3].
Strengthening the gut barrier
As the primary energy source for cells in the intestinal wall, butyrate helps maintain a strong and intact intestinal barrier. This can reduce the risk of the chronic, systemic inflammation associated with cancer development.
Potential in cancer
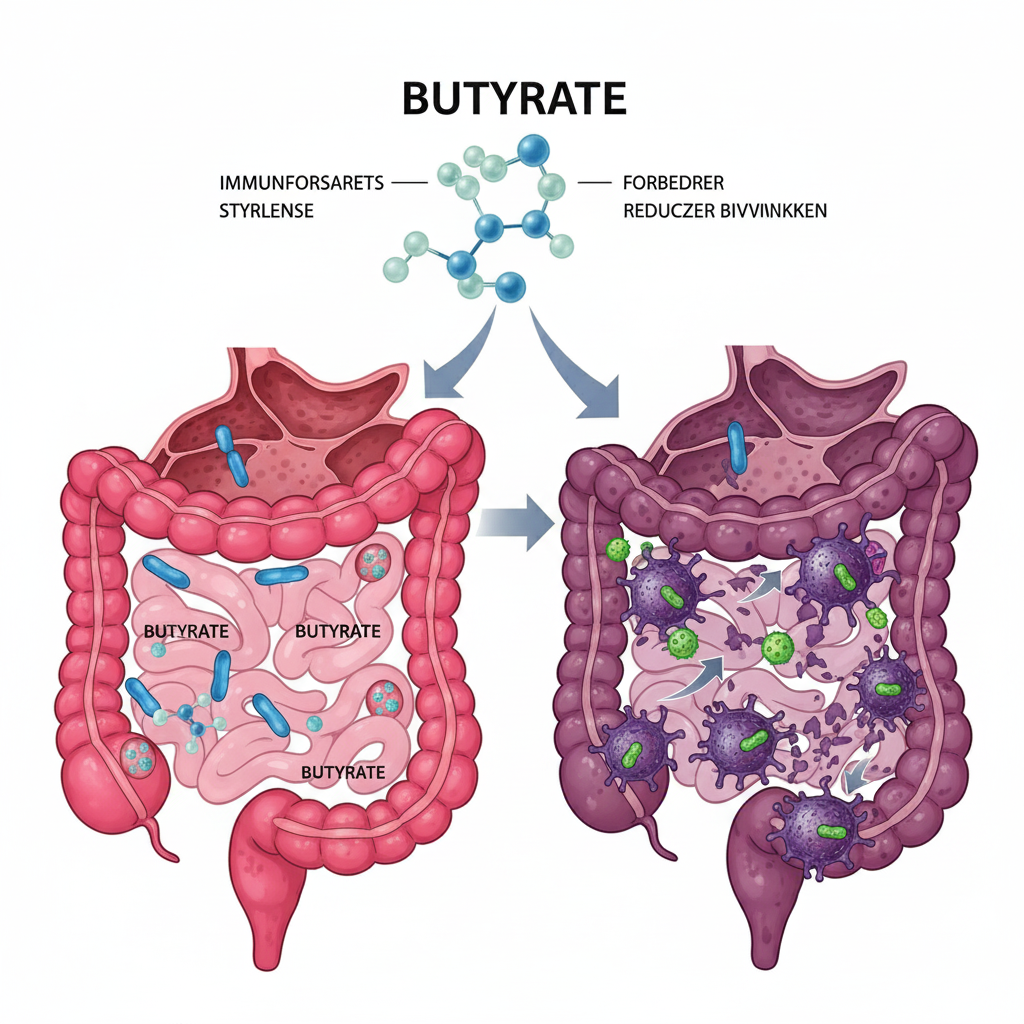
Research into butyrate’s potential as a supplementary treatment for cancer is extensive, particularly in colorectal cancer, but also for other cancer types such as breast, prostate, and lung cancer.
Colorectal cancer
Butyrate’s role is most extensively studied in connection with colorectal cancer, which is logical as this is where the concentration of the substance is highest.
Several studies, primarily in vitro (in test tubes/petri dishes) and in vivo (in living organisms), have shown that butyrate can inhibit the growth of colorectal cancer cells and induce apoptosis. Interestingly, butyrate appears to have a paradoxical effect: it promotes the growth of healthy intestinal cells while inhibiting the growth of cancer cells.
This phenomenon, known as the “butyrate paradox,” makes it particularly promising as a targeted substance that primarily harms malignant cells [4].
Research suggests that this may be due to differences in metabolism between normal cells and cancer cells.
Brain cancer (glioblastoma)
Butyrate’s ability to cross the blood-brain barrier makes it particularly interesting in connection with brain cancer. Research, especially with the more stable derivative phenylbutyrate, has shown that it can inhibit the growth of glioblastoma cells and activate cell death by ‘turning on’ the cancer cell’s silenced defense genes.
Most importantly, studies are investigating its potential to make cancer cells more sensitive to standard treatments such as chemotherapy and radiation, which can enhance their effect [10].
Synergy with conventional treatment
One of the most promising aspects of butyrate is its potential to improve the effect of chemotherapy and immunotherapy.
- Chemotherapy: Studies have shown that butyrate can make cancer cells more sensitive to chemotherapeutic agents such as 5-fluorouracil (5-FU) and oxaliplatin. By opening up the DNA via HDAC inhibition, butyrate can make it easier for these substances to damage the cancer cell’s DNA and thereby increase treatment efficiency [5].
- Immunotherapy: Recent research indicates that the gut flora and its products, including butyrate, play a crucial role in how well immunotherapy works. Butyrate can improve the immune system’s ability to recognize and attack cancer cells. For example, a study from 2023 showed that butyrate can increase the activity of cytotoxic CD8+ T-cells (a type of immune cell) and thereby reinforce the effect of anti-PD-1 immunotherapy, especially in colorectal cancer [6]. It can also counteract cancer cells’ attempts to hide from the immune system by downregulating the PD-L1 protein on the surface of the cancer cells.
Reduction of treatment side effects
Chemotherapy and radiation therapy can cause serious side effects, including mucositis (inflammation and ulceration of the mucous membranes), especially in the gut. By strengthening the intestinal barrier and dampening inflammation, butyrate can potentially reduce these side effects and improve quality of life during treatment.
Benefits of butyrate

In addition to the direct cancer-inhibiting mechanisms, butyrate offers a range of broader health benefits that may be particularly relevant for individuals in cancer treatment.
Improved gut flora
By favoring the growth of beneficial, butyrate-producing bacteria, a fiber-rich diet contributes to a healthier and more robust gut microbiome. A balanced microbiome is associated with better immune function and general health.
Metabolic health
Butyrate can improve insulin sensitivity and has a positive impact on energy metabolism in the body. This can be an advantage, as metabolic syndrome and imbalanced blood sugar can be risk factors for cancer.
Brain protection
There is increasing evidence that butyrate, which as mentioned above can cross the blood-brain barrier, has neuroprotective properties and can support cognitive function.
Disadvantages and limitations
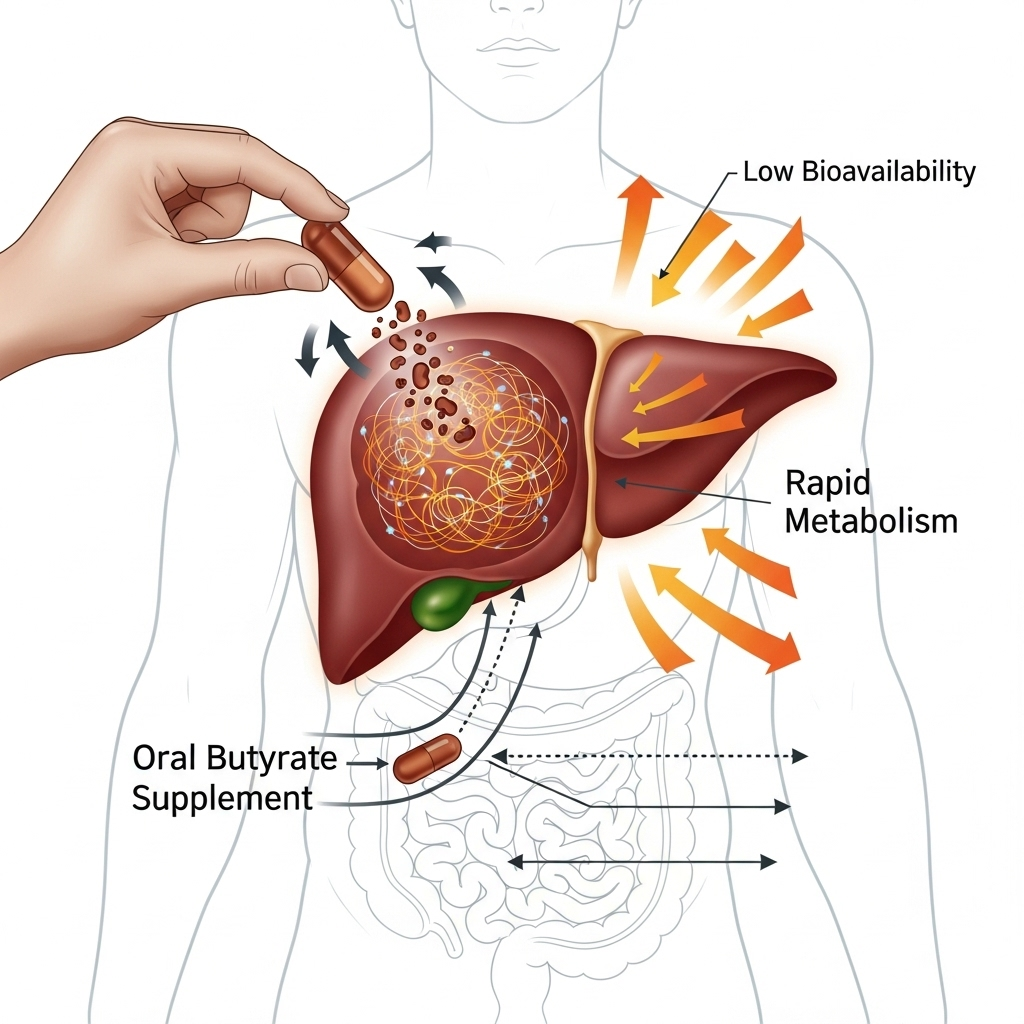
Although the potential is great, there are also challenges and limitations in applying butyrate as a treatment.
Rapid metabolism
When butyrate is taken orally as a supplement, it is absorbed and metabolized very quickly in the gut and liver. This means it is difficult to achieve high and stable concentrations in the blood necessary to influence cancer cells outside the gut.
Bioavailability
Low bioavailability (the proportion of a substance that reaches the bloodstream in active form) is a significant barrier. Researchers are working on developing new methods to deliver butyrate more effectively, for example via pro-drugs (inactive precursors converted to active butyrate in the body) or by encapsulating it so it is released more slowly and further down the intestine.
Need for more human studies
Much of the current knowledge stems from laboratory and animal experiments. Although results are promising, more well-designed clinical trials in humans are needed to establish the real effect, optimal doses, and safety of long-term use as a supplementary cancer treatment.
Clinical trials

The number of clinical trials investigating butyrate directly as a cancer treatment in humans is still limited. Most trials have focused on colorectal cancer and often in combination with other treatments. One example is trials that have investigated the effect of increasing fiber intake or providing fiber supplements to increase natural butyrate production in the gut of individuals with polyps or in high-risk groups for colorectal cancer. The results have generally been positive regarding improving the intestinal environment, but the direct effects on cancer progression in humans are not yet fully clarified [11].
Smaller trials, including Phase I and Phase II studies, have also been conducted where butyrate or its derivatives (e.g., sodium butyrate or phenylbutyrate) have been administered directly, either orally or intravenously.
These trials have shown signs of biological activity, such as increased apoptosis in tumor tissue, but they have also highlighted the challenges of achieving sufficient therapeutic concentrations without side effects [7]. Research is now moving toward more targeted delivery systems and combination trials, which are expected to be tested in future clinical research.
Safety

Butyrate produced naturally in the gut from dietary fiber is considered a completely safe and essential part of the body’s normal function. As a dietary supplement, butyrate salts (e.g., sodium butyrate, calcium butyrate) are generally recognized as safe in moderate doses.
The most common side effects at high doses of oral supplements are mild and related to the gastrointestinal tract, such as bloating or discomfort. Individuals taking the drug sodium phenylbutyrate (a drug metabolized into butyrate) may experience side effects related to the high sodium content.
As with any supplement, one should consult a qualified practitioner before beginning to avoid unwanted interactions, especially if undergoing active cancer treatment.
Dispensing and application

The most effective and natural way to increase butyrate levels is through the diet. Supplements can be an addition but should not replace a healthy diet.
Dietary fiber
Consume a diet rich in fermentable fibers [8]. Good sources include:
- Resistant starch: Cooled cooked potatoes, rice, and pasta, green (unripe) bananas, legumes (beans, lentils, chickpeas), and whole grains like oats and barley.
- Inulin and FOS (fructooligosaccharides): Found in Jerusalem artichokes, garlic, onions, leeks, and asparagus.
- Pectin: Found in fruits such as apples, citrus fruits, and berries.
Butyrate supplements
Supplements are available, most often as salts of butyrate (sodium, potassium, calcium/magnesium butyrate). These are often enteric-coated (with a special film) so they can withstand stomach acid and reach the intestine. Dosage varies but is often between 600 mg and 2000 mg daily, divided into several doses.
Probiotics
Ingestion of probiotics containing butyrate-producing bacterial strains (e.g., Faecalibacterium prausnitzii and Eubacterium rectale) can theoretically help, but it is often more effective to “feed” the existing beneficial bacteria with fibers (prebiotics).
Fiber versus supplements

For cancer patients, especially those prone to inflammation, the question of how best to increase butyrate levels is central. It is important to understand the difference between prebiotic fibers and direct butyric acid supplements:
- Prebiotic fibers (e.g., Psyllium husks/Husk): These are indigestible dietary fibers that function as “food” for the beneficial bacteria in the large intestine. When these bacteria ferment the fibers, they produce butyric acid directly in the gut, where it has the most pronounced anti-inflammatory effect and strengthens the gut barrier. Psyllium husks (“Husk”) are an excellent example of a prebiotic source that can effectively increase your body’s own butyric acid production. This is often the most effective method for achieving high, local concentrations of butyric acid in the gut.
- Butyric acid supplements (capsules/powder): Direct supplements contain pre-made butyric acid (often as sodium butyrate). The challenge is that a significant portion of the butyric acid from oral supplements may be absorbed or metabolized in the upper part of the digestive system or in the liver before reaching the large intestine in sufficient quantities. Although certain products are designed with enteric coating or extended release to improve delivery to the large intestine, research shows that the body’s own production via fiber is often preferable for intestinal health.
For inflammation
In cases of inflammation and to achieve the best results with butyric acid as a supplementary treatment, the most effective strategy is to focus on increasing the intake of fermentable prebiotic fibers. This allows your body to produce butyric acid locally in the large intestine.
Consider supplementing with Psyllium husks (Husk), either as powder or capsules, and prioritize a diet rich in other prebiotic foods as specified above.
Direct butyric acid supplements may be considered as an addition to this strategy but should not replace fundamental fiber intake.
Conclusion
Butyrate is a remarkable molecule formed in the gut as a result of a fiber-rich diet. Its ability to influence cancer cell genes (as an HDAC inhibitor) gives it the potential to slow cell growth, promote cell death, and improve the effect of chemotherapy and immunotherapy.
Although there are challenges with bioavailability regarding direct supplements, current knowledge emphasizes the importance of a healthy gut flora and a fiber-rich diet as a fundamental part of a supportive effort in cancer care [9].
Back to Supplements
Thanks for the tip
To Pia Friis (from the Facebook group I Have Cancer)
Links
[1] The epigenetic effects of butyrate: potential therapeutic implications for clinical practice (PubMed, 2012)
- Content: A scientific review of how butyrate influences gene activity (epigenetics) and the potential for using this in clinical treatment.
[2] Butyrate sensitizes human colon cancer cells to TRAIL-mediated apoptosis (PubMed, 2002)
- Content: A non-randomized laboratory study showing that butyrate can make colorectal cancer cells vulnerable to a signaling substance (TRAIL) that induces them to commit suicide (apoptosis).
[3] Butyrate Suppresses Glucose Metabolism of Colorectal Cancer Cells via GPR109a-AKT Signaling Pathway and Enhances Chemotherapy (PubMed, 2021)
- Content: A non-randomized study showing how butyrate inhibits the sugar metabolism of cancer cells via specific signaling pathways and can simultaneously improve the effect of chemotherapy.
[4] Molecular pathways: gene-environment interactions regulating dietary fiber induction of proliferation and apoptosis via butyrate for cancer prevention (PubMed, 2014)
- Content: A review describing the “butyrate paradox,” where butyrate from dietary fiber can both stimulate growth in healthy cells and promote cell death in cancer cells.
[5] Sodium butyrate in both prevention and supportive treatment of colorectal cancer (PubMed, 2022)
- Content: A recent review article examining the role of butyrate as a supportive treatment in colorectal cancer, including its interaction with chemotherapy.
[6] Gut microbial metabolites facilitate anticancer therapy efficacy by modulating cytotoxic CD8+ T cell immunity (PubMed, 2021)
- Content: A non-randomized study showing how gut bacteria products, including butyrate, can strengthen the immune system’s killer cells (CD8+ T-cells) and thereby improve the effect of cancer treatment.
[7] Combination of phenylbutyrate and 13-cis retinoic acid inhibits prostate tumor growth and angiogenesis (PubMed, 2001)
- Content: A non-randomized laboratory study demonstrating that a butyrate-like substance in combination with vitamin A can inhibit the growth of prostate cancer and the formation of new blood vessels for the tumor.
[8] Få den gode effekt af fiber i kosten – men slip for at spise fiber (DCA, Nationalt Center for Fødevarer og Jordbrug, 2014)
- Content: A Danish article conveying research on how to achieve the health benefits of butyrate produced from fibers.
[9] Butyrate as a promising therapeutic target in cancer: From pathogenesis to clinic (Review) (PubMed, 2024)
- Content: A 2024 review article summarizing the latest knowledge on butyrate as a promising agent in cancer treatment, from basic research to clinical application.
[10] Recent developments of HDAC inhibitors: Emerging indications and novel molecules (British Pharmacological Society, 2021)
- Content: A 2021 review article detailing recent developments and clinical trials with HDAC inhibitors (including substances like phenylbutyrate) for the treatment of cancers such as glioblastoma.
[11] Cancer Prevention with Resistant Starch in Lynch Syndrome Patients in the CAPP2-Randomized Placebo Controlled Trial: Planned 10-Year Follow-up (PubMed, 2022)
- Content: A large-scale randomized placebo-controlled clinical trial showing that resistant starch (a potent fiber for butyric acid production) did not reduce the incidence of colorectal cancer but significantly reduced other cancer types in a high-risk group.
Page created: August 29, 2025.
❤
What you read on I Have Cancer is not a recommendation. Seek competent guidance.

